
MDSAP Quality Certification Awarded to Jointechlabs
Jointechlabs earned the MDSAP quality certification indicating compliance with ISO13485:2016 for medical device quality management.
Read more
Jointechlabs earned the MDSAP quality certification indicating compliance with ISO13485:2016 for medical device quality management.
Read more
European focus includes a new subsidiary in Austria, a medical research center in partnership with a prominent health center, and development and distribution agreements for Germany, Austria, Switzerland, Italy, Spain, and the Balkans.
Read more
Jointechlabs has just strengthened its intellectual property position with the granting of a new US patent covering systems and methods of encapsulating stem cells.
Read more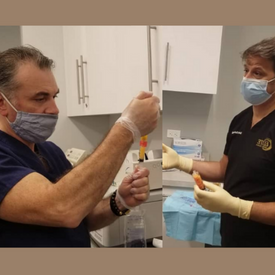
Drs. Katz and Guarino demonstrate the operation of MiniTC.
Read more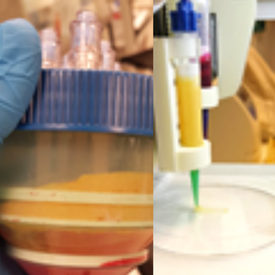
Critical skin wounds are a major cost for the health sector and lead to immense suffering for the patients. One way to facilitate the healing of these wounds is through autologous adipose tissue transplants. However, the implanted tissue needs to be vascularized or it will suffer from necrosis. To combat this Stromal vascular fraction (SVF), […]
Read more
Treating patients with OA presents a significant challenge for physicians as no therapies to date have demonstrated efficacy in curing or even halting disease progression. Therefore, most approaches initially target pain management and factors that may be exacerbating stress on the joint. https://dx.doi.org/10.2147%2FSCCAA.S68073 – Mayo Clinic, MN Corticosteroid toxicity for chondrocytes has been long reported, both […]
Read more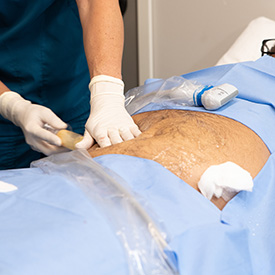
Katz, N., Koukharenko, V. and Geldner, P2,3. Laboratory of Stem cells and Bioengineering, Jointechlabs, Inc., Skokie, IL North Michigan Surgical Center, Chicago, IL Geldner Center, Chicago, IL Introduction: Fat auto graft transplantation method for different reconstructive plastic surgeries has been fueled recently by encouraging reports from Japan, the United Kingdom, the United States, and others. […]
Read more
Jointechlabs Launches MiniTC for Point-of-Care Fat Tissue Processing The scientific community has regarded regeneration as a topic of interest for thousands of years. Long before it was dubbed ‘Regenerative Medicine’, this type of medical intervention was applauded by healthcare professionals for its successful outcomes. Early discoveries, such as skin graft procedures for facial reconstructions, have […]
Read more
Jointechlabs – a leader in point-of-care regenerative medicine therapies – today announced that the U.S. Food and Drug Administration (FDA) cleared the company’s MiniTCTM for point-of-care fat tissue processing designed to obtain microfat (or fat grafts), for multiple indications. Jointechlabs plans to focus on a range of therapeutic areas including medical aesthetics, plastic surgery, orthobiologics […]
Read more